Just because a diet is popular does not mean that it lives up to its claims nor does it mean that it’s safe! Case in level: ketogenic diets. I have been a loud voice of dissent within the Paleo and alternative health communities for about the last five years, conveying my deep concern about the documented inherent perils of obeying a ketogenic diet, while supporters issue statements that amount to little more than propaganda, expounding on the potential benefits yet rejecting the immense quantity of scientific indicating that attracts those results into question and that explains the mechanisms behind how ketogenic diets can cause harm. In reality, ketogenic diets absolutely do present therapeutic potential for some medical precondition( such as refractory epilepsy and multiple sclerosis ), but the choice to follow a ketogenic diet requires an informed and careful cost-benefit analysis and close medical supervision.
Just because a diet is popular does not mean that it lives up to its claims nor does it mean that it’s safe! Case in level: ketogenic diets. I have been a loud voice of dissent within the Paleo and alternative health communities for about the last five years, conveying my deep concern about the documented inherent perils of obeying a ketogenic diet, while supporters issue statements that amount to little more than propaganda, expounding on the potential benefits yet rejecting the immense quantity of scientific indicating that attracts those results into question and that explains the mechanisms behind how ketogenic diets can cause harm. In reality, ketogenic diets absolutely do present therapeutic potential for some medical precondition( such as refractory epilepsy and multiple sclerosis ), but the choice to follow a ketogenic diet requires an informed and careful cost-benefit analysis and close medical supervision.
Today’s post is not designed to relitigate this debate, but I do spur you to read Adverse Reactions to Ketogenic Diet: Caution Advised. Instead, I had wished to dive deep into one of the risks that ketogenic diets pose that I have not yet discussed in detail: the impact of keto on the gut microbiome!
Up until recently, experiment on how ketogenic diets impact the human bowel microbiota has been extremely limited, have contributed to lots of speculation without much hard data. Luckily, in the last few years alone, that’s modified! Several learns have come out examining the intestine microbiota effects of ketogenic diets, helping us paint a picture of what really happens in our bowel when we go keto. And, the findings raise some major red flags.
What Does the Science Show?
First, it’s worth emphasizing that the composition of our gut microbiome alters every structure, and indeed every cell, in our bodies. We benefit greatly when we have a diverse gut microbiome, abundant in key protiotic species. See What Is the Gut Microbiome? And Why Should We Care About It ?. I am currently writing an entire book on the best meat to eat to support a health, diverse gut microbiome, but the quick summary is: abundance of high-fiber and phytonutrient-rich fruits and vegetables( aiming for at 8+ provides of veggies daily and ideally 45-50 grams of fiber) that reach as many fresh fruit and vegetable groups daily as possible–cruciferous vegetables, mushrooms, roots, tubers, alliums, leafy dark-greens, berries, apple family, citrus–as well as nuts and seeds, herbs and spices, peas, extra innocent olive oil, fish, shellfish, honey and bee products, fermented foods, edible bugs, tea, coffee, cocoa and bone broth. See also Why Root Veggies Are important for the Gut Microbiome, Why Fish is Great for the Gut Microbiome, Honey: The Sweet Truth About a Functional Food ! and watch my recent Paleo f( x) 2019 talk on the microbiome here.
Given how incongruous the tenets of a ketogenic diet are with the above principle for microbiome health, it shouldn’t come as a amaze that ketogenic diets can dramatically impact the bowel. We know from existing experiment that notable increases in fat and reductions in fermentable substrate( like fiber and resistant starch) alter the intestinal surrounding and vary the food supply reaching our occupant microbes( watch also Saturated Fat: Healthful, Harmful, or Somewhere In Between ?, Resistant Starch: It’s Not All Sunshine and Roses, and The Fiber Manifesto-Part 1 of 5: What Is Fiber and Why Is it Good? ). Interestingly, for some of the conditions keto has been shown to benefit( such as epilepsy and autism ), the effects are at least partially mediated through intestine bacteria. For example, in a mouse model of autism, a ketogenic diet had a distinct antimicrobial influence( causing a significant drop in bacterial abundance) and helped reduce elevated levels of Akkermansia muciniphila, who the hell is abnormally high among the BTBR mice relative to the self-control swine. Although lower bacterial tiers and a decrease in beneficial Akkermansia muciniphila aren’t typically considered desirable for bowel health, these changes are is connected with improvements in autism symptoms. We are also aware that in humans, certain antibiotics( such as vancomycin) have all contributed to reduced autism symptoms in children around the disease, recommending that the antimicrobial effect of ketogenic diets could simulate that of antibiotics and improve symptoms of autism through similar mechanisms.
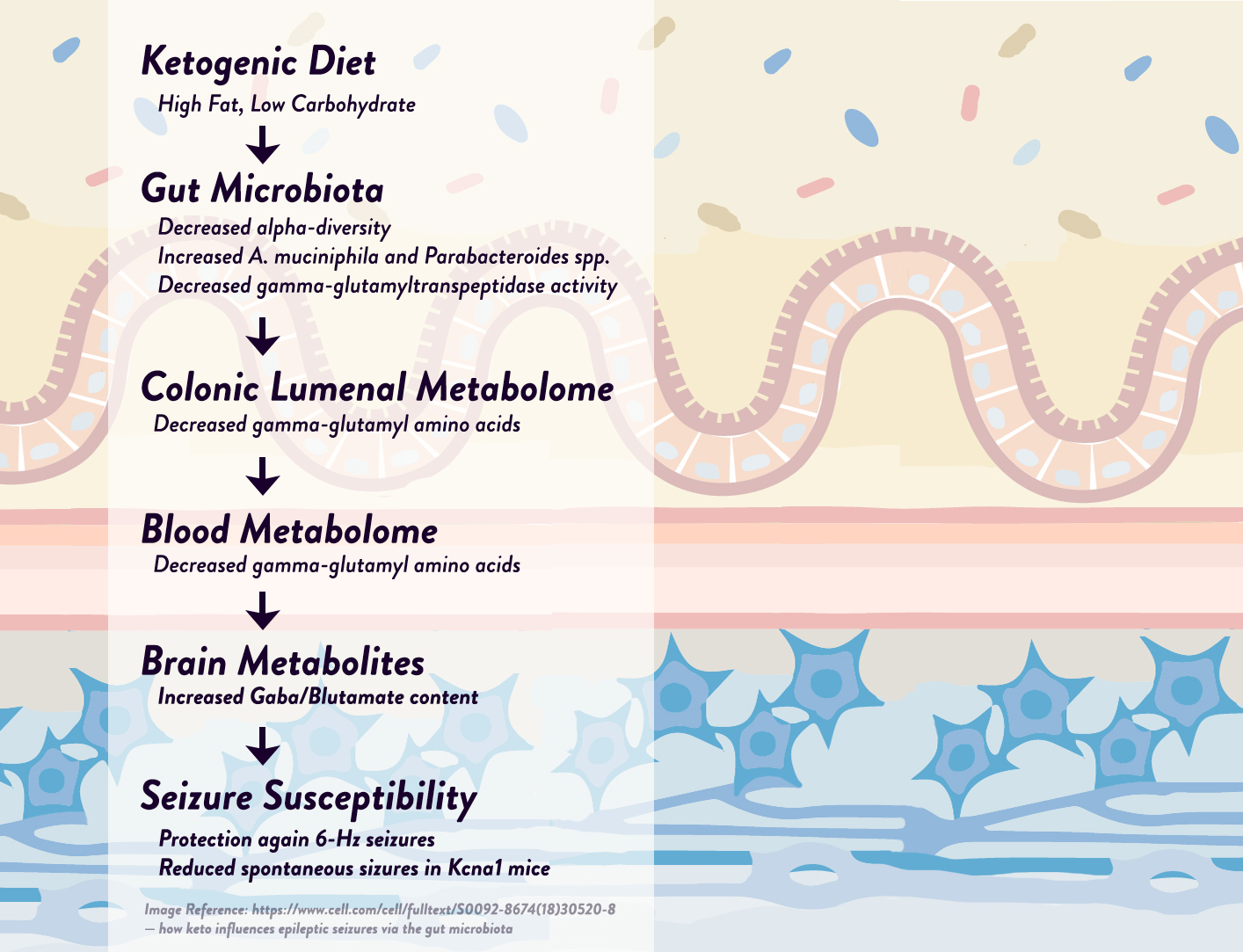
Likewise, utilizing a mouse modeling of epilepsy, researchers discovered that two specific bacteria–Akkermansia muciniphila and Parabacteroides–were elevated by the ketogenic diet and showed held accountable for the diet’s anti-epileptic influences, working synergistically to affect neurotransmitter activity in the hippocampus( the part of the brain that generates many epileptic seizures). More specifically, these two bacteria together–but not individually–were able to reduce levels of gamma-glutamylated amino acids in the gut and blood, in turn raise hippocampal GABA/glutamate grades and reducing the frequency of convulsions in the mice. As indicating that the microbiota modifications held accountable for the convulsion reduction, health researchers tested the ketogenic diet on mice with sterilized microbiomes( either from antibiotic therapy or from being reared in a germ-free environment) and found that the diet was no longer protective against seizures! Although more studies be necessary for humen, we is well aware from earlier experiment that epileptic infants respond to ketogenic diets with a decrease in seizure frequency and an increase in Prevotella and Bifidobacterium populations, supporting the idea that specific bacteria has been possible to anti-seizure influences in humans as well as mice.
But, while ketogenic-diet-induced changes in the gut microbiota can benefit very specific medical conditions, the overall impact on gut health isn’t so rosy-cheeked. As we are only touched upon, the consequences of ketogenic diets on epilepsy–consistent throughout multiple studies–includes a drop in bacterial diversity, a near-universal hallmark for a less-healthy, less-resilient microbiome. Here’s what the other existing literature has to say!
In a study of 12 children with therapy-resistant epilepsy, three months of ingesting a ketogenic diet resulted in a significant drop in some important bacteria groups, including Bifidobacterium( a genus of probiotics that produce vitamins, regulate the gut’s microbial homeostasis, help prevent pathogens from infecting the gut, boost immune function, and much more !). Bifidobacteria grades plummeted from an average relative abundance of fifteen. 8% before the ketogenic diet to only 3.9% after three months. And, two species in particular take a big hit: Bifidobacterium longum( which assistances mediate immune function and promotes good digestion) dropped 3.3-fold, and Bifidobacterium adolescentis( which synthesizes vitamin B12, thiamin, vitamin B6, and folic acid) fell 16 -fold. Likewise, the ketogenic diet been successful in lower levels of Eubacterium rectale( a major producer of short-chain fatty acids) and an increased abundance of Escherichia coli( which includes several pathogenic stress famous for make food poisoning, and whose overgrowth has been linked to Crohn’s disease ).
In this study, the researchers noted that the significant drop in Bifidobacterium and Eubacterium rectale could result in lower different levels of the short-chain fatty acids acetate and lactate( which are products of the bifid shunt–the unique fermentative pathway used by bifidobacteria ), in turn reducing the acidity of the intestine. Maintaining a low-spirited pH in the gut is one of the ways our body assists avoid intestinal infection with pathogens( many of which are acid-sensitive), and this could explain why the compositional changes induced by a ketogenic diet included an increase in E. coli abundance.
In another examine, researchers looked at the effects of a ketogenic diet on patients with Glucose Transporter 1 Deficiency Syndrome( GLUT1-DS ), a rare metabolic disorder that avoids glucose from bridging the blood-brain barrier. Along with consuming a classical ketogenic diet( a 4:1 rate of fat grams to carbohydrate plus protein grams ), the participants “il give” vitamin and mineral supplements to prevent micronutrient deficiencies, which can independently alter the intestine microbiota composition. After three months eating keto, the biggest change in the participants’ microbiota was a significant rise in Desulfovibrio, a genus of sulfate-reducing bacteria that’s been found in relatively high concentrations in people with inflammatory bowel infection.( Desulfovibrio can cause inflammation in the gut mucosa by activating immune the replies and make hydrogen sulfide, which acts as a pro-oxidant and can be toxic to cells!)
Intriguingly, the increases in Desulfovibrio may be specifically linked to increased intake of dairy fat, which moved up substantially for most participants in this study( median saturated fat intake increased from 8.4% of calories at baseline to 23.5% on the diet, largely from butter, ointment, cheese, and other high-fat dairy foods ). In mice, researchers have shown that saturated fat from dairy( but not polyunsaturated fat from safflower oil) promotes the taurine conjugation of bile acids, which in turn increases the availability of organic sulfur that bacteria like Desulfovibrio use. These varied bowel circumstances produce to an expansion of sulfate-reducing microbes, and a potentially higher peril of inflammatory bowel ailments. So, when it is necessary to ketogenic diets, milk-derived flabs may persuade a shift in bile battery-acid composition that then promotes the growth of Desulfovibrio! We’ll need more studies to determine whether other sources of saturated fat have a similar effect.
 Interestingly, a longer-term survey in patients with MS showed something unique among the available literature: an initial drop in microbiota response to ketogenic eating, followed by a convalescence to normal levels. In this study, MS patients were put on diets designed to achieve modest ketosis( at the least 500 mmol/ L ss-hydroxybutyrate in the blood and >=500 mmol/L acetoacetate in the urine) for a total of six months.( This diet was only 69% calories from fat( 21% from protein and 10% from carbohydrates) which is on the cusp for achieving sustained ketosis, compared to “classic” ketogenic diets having 75-80% calories from fat ).
Interestingly, a longer-term survey in patients with MS showed something unique among the available literature: an initial drop in microbiota response to ketogenic eating, followed by a convalescence to normal levels. In this study, MS patients were put on diets designed to achieve modest ketosis( at the least 500 mmol/ L ss-hydroxybutyrate in the blood and >=500 mmol/L acetoacetate in the urine) for a total of six months.( This diet was only 69% calories from fat( 21% from protein and 10% from carbohydrates) which is on the cusp for achieving sustained ketosis, compared to “classic” ketogenic diets having 75-80% calories from fat ).
Up until the 12 th week of the study, all groups of bacteria other than Akkermansia decreased, leading to significant drop in total bacterial concentration and a decrease in bacterial diversity from 48% to 35%( with some groups of formerly present bacteria falling below the detecting level ). The quantity of these changes were similar to the effects of antibiotics! Around week 12 of such studies, though, bacterial concentrations started to rise again, and near the six-month mark, they were significantly higher than the concentrations the MS patients had at baseline( and were similar to the average bacterial concentrations seen in healthy controls).
So, does this mean ketogenic diets will reinstate a healthy microbiome as long as we stick to them long enough? Not so fast! MS patient started out with unique microbiomes( low-spirited in biofermentative bacteria, relative to health powers ), making it hard to extrapolate the findings and conclusions to other groups of people, and the study was relatively small( merely 10 players had their microbiota analyzed). Likewise, this study was relatively higher in protein and carbohydrate( 150 grams per day blended) than classical ketogenic diets with a higher proportion of fat, who are capable of potentially play important roles in the results. What’s more, studies of similar segment with epileptic patients didn’t ensure a recovery in concentration or diversity after to put into practice ketogenic diets for longer periods of time. At best, this study suggests that ketogenic diet may be specifically helpful to MS patients, but we can’t assume everyone else will be so lucky!
What about for people who don’t have specific medical conditions that keto is known to benefit? We are able to obtain some evidences about this by looking at weight-loss examines. One trial placed participants on two different weight-loss diets lasting four weeks each( a moderate carbohydrate diet with 164 grams of carbohydrate per day, and a ketogenic diet with 24 grams of carbohydrate per day), and found that the ketogenic diet stage brought the greatest reduced by the Roseburia and Eubacterium rectale group, which plays a key role in producing butyrate and the protection colonic health. Likewise, bifidobacteria grades decreased more dramatically when participates were eating the ketogenic diet versus the moderate carbohydrate diet. The preponderance of prove indicates such changes are a net negative.
What Do We Make of it All?
Although more research is definitely needed to clarify( and expand on !) the findings we currently have, there are a few observations we can induce so far.
First, the good news. For certain medical conditions, specially autism and epilepsy and potentially MS, the effects of a ketogenic diet on the microbiota composition seem to be beneficial–at least for the condition in question. Even when alters pass that are typically considered detrimental, such as a drop in diversity, other modifies seem to specifically mediate pathways that contribute to disease, stimulating the pros arguably outweigh the cons.
 But, for people who aren’t treating a particular medical condition that ketogenic diets have already been shown to benefit, there’s virtually no evidence that running keto will do your microbiota any favors. The advertising of a less acidic colonic environ( due to reduced fermentation of plant-derived substrates, and subsequently a lower production of short-chain fatty battery-acids) has the potential to be an issue in long-term ketogenic diets. According to Jeff Leach, microbiome researcher and founder of the Human Food Project, a shift in pH and available substrate develops environmental issues where acid-sensitive pathogens can flourish( including members of Escherichia, Salmonella, Helicobacter, and Vibrio ), while at the same time starvation important butyrate producers like Eubacterium and Roseburia. Combine with the effects of a higher saturated fat intake on gut roadblock soundnes, the microbial community shaped by ketogenic diets is likely to increase bowel permeability and ultimately shifting the microbiome to a country of dysbiosis. The promotion of sulfate-reducing bacteria in the gut is a serious concern, especially with ketogenic diets that rely heavily on dairy flabs, since hydrogen sulfide( the final product of sulfate-reducing bacteria like Desulfovibrio) is known to harm the intestine mucosa and promote inflammation through multiple mechanisms. We are also aware that when intestine bacteria have a short supply of carbohydrate, they turn to alternative energy sources like amino battery-acids, causing them to produce metabolites that may be harmful to human health.
But, for people who aren’t treating a particular medical condition that ketogenic diets have already been shown to benefit, there’s virtually no evidence that running keto will do your microbiota any favors. The advertising of a less acidic colonic environ( due to reduced fermentation of plant-derived substrates, and subsequently a lower production of short-chain fatty battery-acids) has the potential to be an issue in long-term ketogenic diets. According to Jeff Leach, microbiome researcher and founder of the Human Food Project, a shift in pH and available substrate develops environmental issues where acid-sensitive pathogens can flourish( including members of Escherichia, Salmonella, Helicobacter, and Vibrio ), while at the same time starvation important butyrate producers like Eubacterium and Roseburia. Combine with the effects of a higher saturated fat intake on gut roadblock soundnes, the microbial community shaped by ketogenic diets is likely to increase bowel permeability and ultimately shifting the microbiome to a country of dysbiosis. The promotion of sulfate-reducing bacteria in the gut is a serious concern, especially with ketogenic diets that rely heavily on dairy flabs, since hydrogen sulfide( the final product of sulfate-reducing bacteria like Desulfovibrio) is known to harm the intestine mucosa and promote inflammation through multiple mechanisms. We are also aware that when intestine bacteria have a short supply of carbohydrate, they turn to alternative energy sources like amino battery-acids, causing them to produce metabolites that may be harmful to human health.
The evidence we have so far suggests that drastically decreasing carbohydrate uptake( specially fiber and resistant starch) will reduce populations of some very important bacteria, and a concurrent increase in flab( specially saturated fat) can alter systemic endotoxin levels, intestinal permeability, inflaming, and microbiota composition. Collectively, these components that define ketogenic diets seem to be a lethal combination for our gut health.
 This isn’t to say that ketogenic diets don’t have very real, significant benefits for many people, or that everyone who runs keto will automatically face severe bowel health issues. However, being realistic about the potential trauma that ketogenic diets can bring is important in order to safeguard against future difficulties( again I foster you to read Adverse Reactions to Ketogenic Diet: Caution Advised ). Supplementing with prebiotic foods and potentially certain probiotic bacteria, ingesting a diverse array of fibrous plant foods, preserving saturated fat uptake( especially dairy fat) at reasonable tiers( discover Saturated Fat: Healthful, Harmful, or Somewhere In Between ? ), and taking other measures to optimize gut health( including lifestyle factors like adequate sleep, stress management, and healthy activity levels, recognize The Paleo Lifestyle) may help take the edge off some of the gut microbiota risks involved with ketogenic eating. But, very good solution for many people may simply be eating a nutrient-dense diet that retains a higher level of fiber- and phytochemical-rich carbohydrate meat to support a healthy microbial community in our gut( understand The Importance of Vegetables and The Case for More Carbs: Insulin’s Non-Metabolic Roles in the Human Body ).
This isn’t to say that ketogenic diets don’t have very real, significant benefits for many people, or that everyone who runs keto will automatically face severe bowel health issues. However, being realistic about the potential trauma that ketogenic diets can bring is important in order to safeguard against future difficulties( again I foster you to read Adverse Reactions to Ketogenic Diet: Caution Advised ). Supplementing with prebiotic foods and potentially certain probiotic bacteria, ingesting a diverse array of fibrous plant foods, preserving saturated fat uptake( especially dairy fat) at reasonable tiers( discover Saturated Fat: Healthful, Harmful, or Somewhere In Between ? ), and taking other measures to optimize gut health( including lifestyle factors like adequate sleep, stress management, and healthy activity levels, recognize The Paleo Lifestyle) may help take the edge off some of the gut microbiota risks involved with ketogenic eating. But, very good solution for many people may simply be eating a nutrient-dense diet that retains a higher level of fiber- and phytochemical-rich carbohydrate meat to support a healthy microbial community in our gut( understand The Importance of Vegetables and The Case for More Carbs: Insulin’s Non-Metabolic Roles in the Human Body ).
Citations
Brinkworth GD, et al. “Comparative influences of a very low carbohydrate, high-pitched fat and high-carbohydrate, low-fat weight loss diets on bowel habits and feacal short chain fatty acid and bacterial populations.” British J Nutri. 2009;201(10):1493-1502.
Devkota S, et al. “Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10-/- mouse.” Nature. 2012 Jul 5; 487( 7405 ): 104 -8. doi: 10.1038/nature11225.
Earley H, et al. “A Preliminary Study Examining the Binding Capacity of Akkermansia muciniphila and Desulfovibrio spp ., to Colonic Mucin in Health and Ulcerative Colitis.” PLoS One. 2015 Oct 22; 10( 10 ): e0135280. doi: 10.1371/ journal.pone. 0135280. eCollection 2015.
Ellerbroek, A. “The effect of ketogenic diets on the bowel microbiota.” Journal of Exercise and Nutrition. 2018; 1( 5 ).
Leach, J. “Sorry Low Carbers, Your Microbiome is Just Not That Into You.” Human Food Project. 26 June 2013. http://humanfoodproject.com/sorry-low-carbers-your-microbiome-is-just-not-that-into-you/
Lindefelt M, et al. “The ketogenic diet influences taxonomic and functional composition of the bowel microbiota in children with severe epilepsy.” NPJ Biofilms Microbiomes. 2019 Jan 23; 5:5…
Read more: thepaleomom.com
 iSmartNetwork Magazine The online magazine for you…
iSmartNetwork Magazine The online magazine for you…